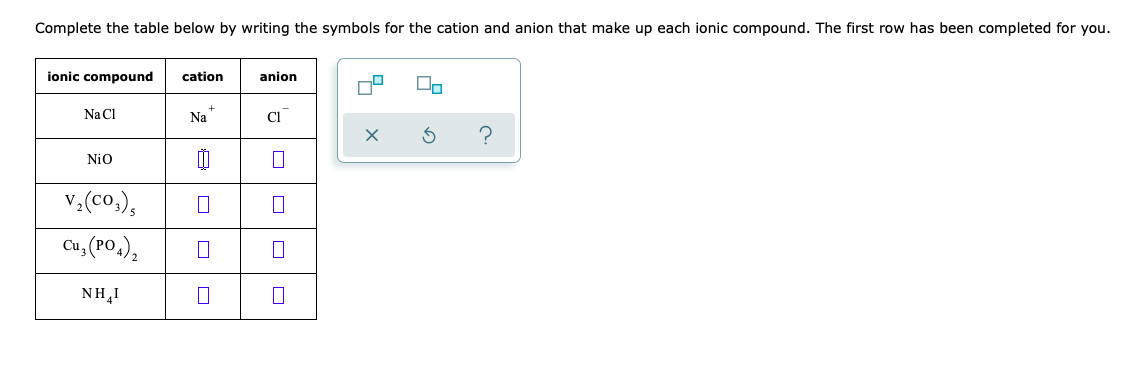
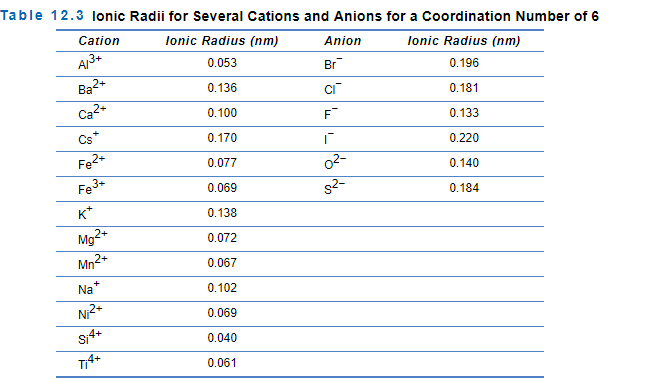
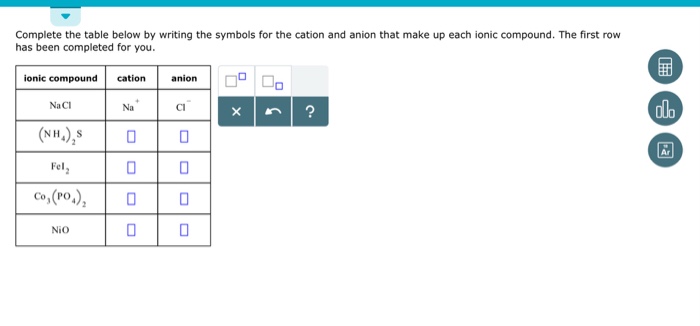
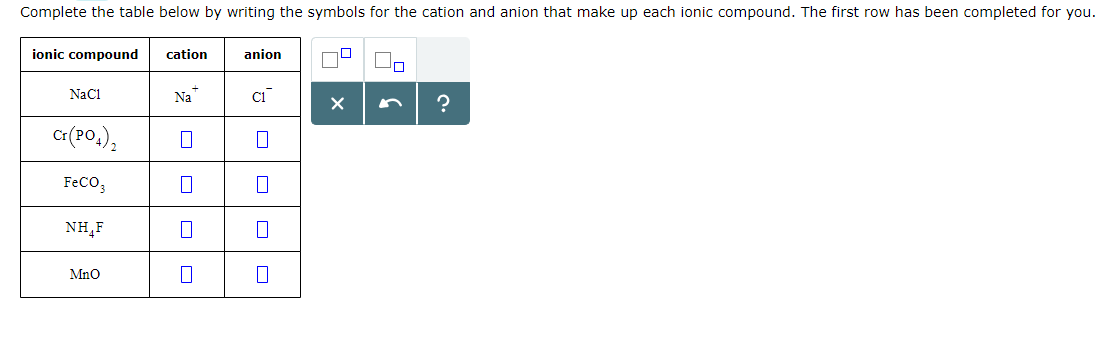
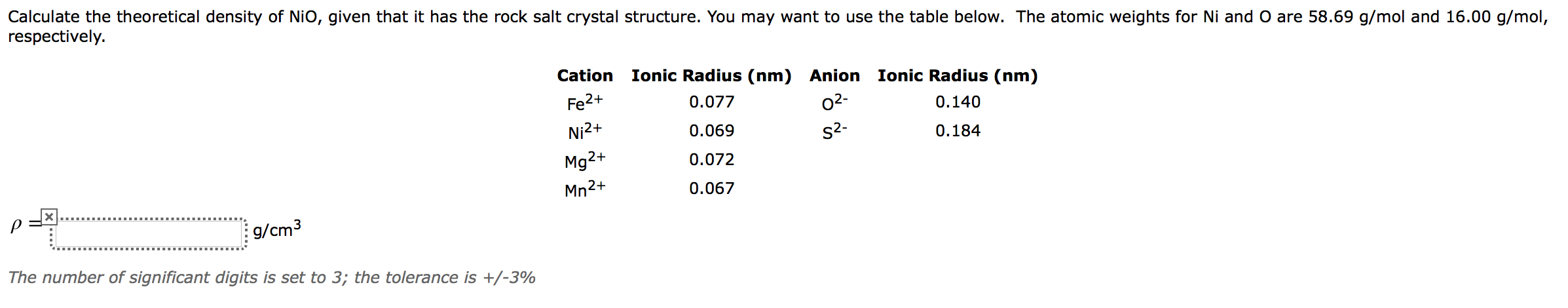
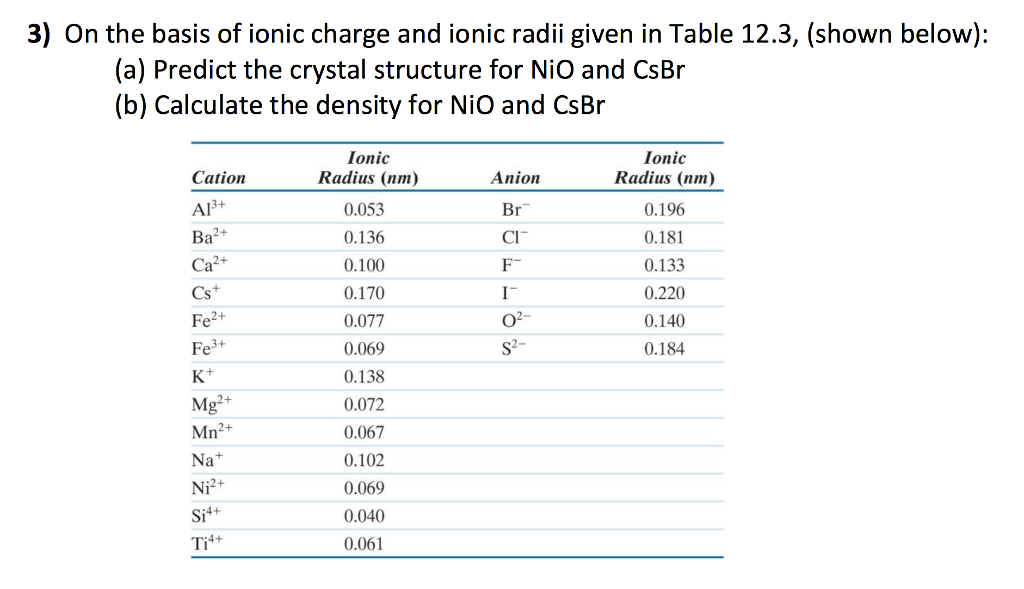
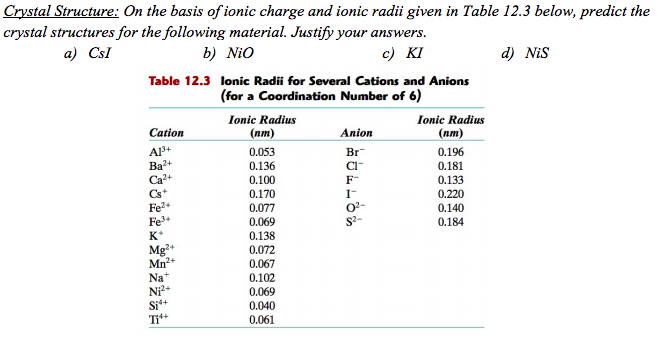
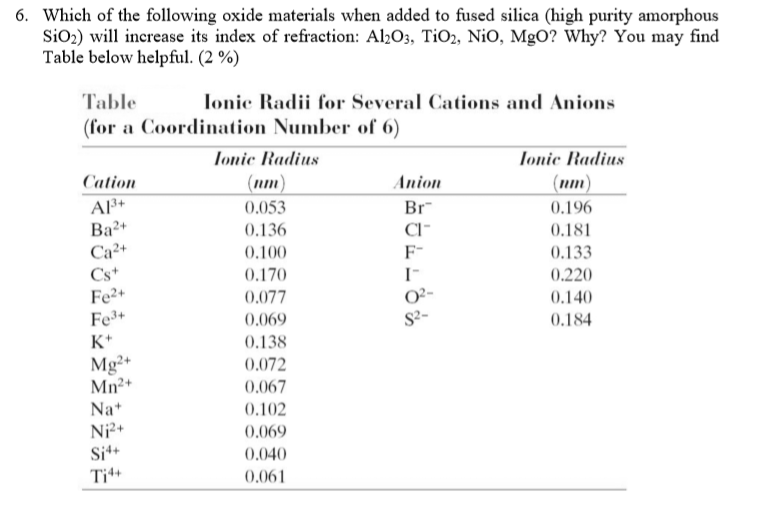
Nio Cation And Anion
Ions are atoms or molecules which have gained or lost one or more valence electrons giving the ion a net positive or negative charge.
Nio cation and anion. Yahia djaoued electrochromic and colorimetric properties of anodic nio thin films. A good way to remember which type of ion is positively charged is to use the following mnemonic devices. Such atoms are referred to as ions. Draganjac s introduction to chemistry chem1003 general chemistry i chem1013 and general chemistry ii chem1023 classes are responsible for learning the names and formulae for the common acids and common reagents and for learning the names formulae and the charges for the common cations and anions listed below.
Current location formulas in chemistry chemistry basics cations and anions. Depending upon the charge it acquires an ion is classified into two types cation and anion. A cation is an atom which has lost one or more electrons. Large sized anions occupy lattice sites while small sized cations are found in voids.
The ratio of radius of cation to anion is called radius ratio. Cations positively charged ions and anions negatively charged ions are formed when a metal loses electrons and a nonmetal gains those electrons. The difference between a cation and an anion is the net electrical charge of the ion. Several million kilograms are produced annually of varying quality mainly as an intermediate in the production of nickel alloys.
Anion originated from the greek word ano meaning up the word cation originated from its greek counterpart kata which means down. A metal reacts with a nonmetal to form an ionic bond. Cation anion based electrochemical degradation and rejuvenation of electrochromic nickel oxide thin films. Cations and anions are both ions.
In condensed matter physics and inorganic chemistry the cation anion radius ratio also. Ths different ions combine together to form molecules and the. And the ions with a negative charge on them are called anions. Nickel ii oxide is the chemical compound with the formula nio it is the principal oxide of nickel.
The electrostatic attraction between the positives and negatives brings the particles together and creates an ionic compound such as sodium chloride. Cation anion list worksheet for naming ions students enrolled in dr. The mineralogical form of nio bunsenite is very rare other nickel iii oxides have been claimed for example. Don t forget to try our free app agile log which helps you track your time spent on various projects and tasks try it now.
Radius ratio rule 1 is the ratio of the ionic radius of the cation to the ionic radius of the anion in a cation anion compound. You can often. It is classified as a basic metal oxide.