Nio Band Structure
Nio is halite rock salt structured and crystallizes in the cubic fm 3m space group.
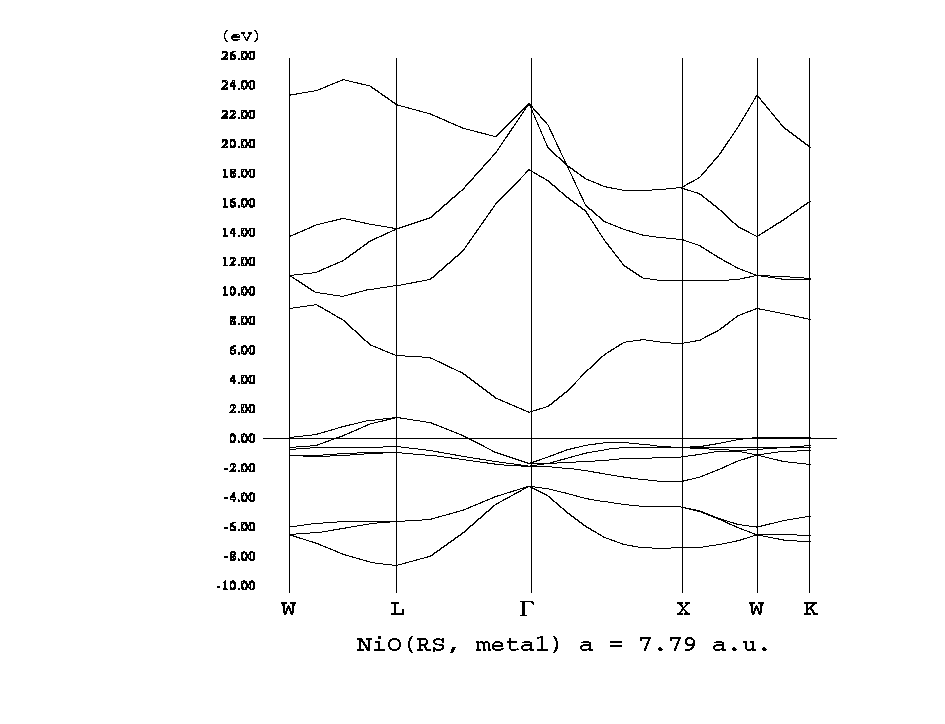
Nio band structure. Ni2 is bonded in a linear geometry to two equivalent o2 atoms. Ni2 is bonded to six equivalent o2 atoms to form a mixture of corner and edge sharing nio6 octahedra. The band structure of nio is being the ob ject of many calculations by different techni ques. Nio adopts the nacl structure with octahedral ni 2 and o 2 sites.
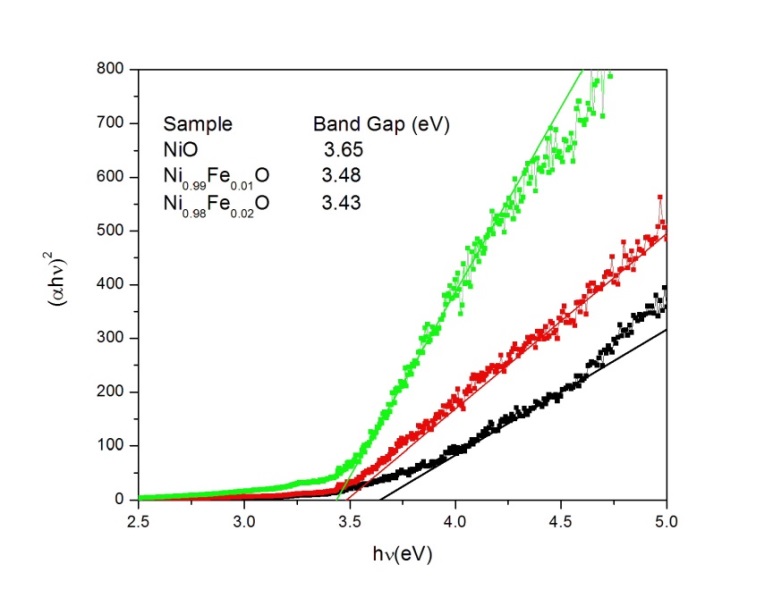
Most authors using computational techniques well beyond the simple density functional theory and the approximations gga or lda claim that the band gap is about 4 0 ev and that the conduction band is of ni 3d nature. The conceptually simple structure is commonly known as the rock salt structure. The corner sharing octahedral tilt angles are 0. The band structure of the prototypical charge transfer insulator nio is computed by using a combi nation of an ab initio band structure method and the dynamical mean field theory with a quantum monte carlo impurity solver.
Nio crystallizes in the orthorhombic cmmm space group. All ni o bond lengths are 2 10 å. O2 is bonded in a linear geometry to two equivalent ni2 atoms. Although argu ably the most studied and modeled of binary transition metal oxides exact details of the band structure band gap e g fermi level e f conduction band minimum cbm valence band maximum vbm and conduction mechanism continue to stimulate discussion 39 43 45.
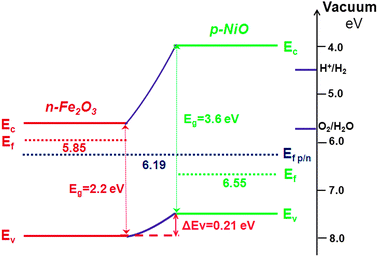
This model have been successfully applied to describe many details of electronic structure magnetic coupling and character of the band gap since a long time. In this tutorial you will compare the dft and dft u models for this system using the gga. The band structure of a strongly correlated semiconductor as nio has been the object of much debate prl 103 036404 2009. Nio was chosen as the p type interfacial layer.
The nio crystal has a too low band gap in lda and gga and is one of the standard examples of how the dft u approximation can be used to improve the description of the electronic structure of solids. The structure is three dimensional. The ni atoms have a net magnetic moment and form an anti ferromagnetic arrangement in the 111 direction of the fcc cell. Employing a hamiltonian which includes both ni d and o p orbitals we find.
Thus they seem to forget the. Like many other binary metal oxides nio is often non stoichiometric meaning that the ni o ratio deviates from 1 1. The structure can be described by a rhombohedral unit cell with 4 atoms in the basis cdg05. Nio has a fcc crystal structure with two atoms in the unit cell.
The structure is one dimensional and consists of two nio ribbons oriented in the 0 0 1 direction. Both ni o bond lengths are 1 72 å. To quote just a few physical review letters recently published the works of engel and schmid 1 on.